How To Draw Crystal Field Splitting Diagram
Crystal Field Theory
- Page ID
- 529
Crystal field theory (CFT) describes the breaking of orbital degeneracy in transition metal complexes due to the presence of ligands. CFT qualitatively describes the strength of the metal-ligand bonds. Based on the strength of the metal-ligand bonds, the energy of the system is altered. This may atomic number 82 to a change in magnetic properties also as colour. This theory was developed by Hans Bethe and John Hasbrouck van Vleck.
Basic Concept
In Crystal Field Theory, it is assumed that the ions are elementary point charges (a simplification). When applied to alkali metallic ions containing a symmetric sphere of charge, calculations of bond energies are by and large quite successful. The approach taken uses classical potential energy equations that have into account the attractive and repulsive interactions between charged particles (that is, Coulomb's Police force interactions).
\[E \propto \dfrac{q_1 q_2}{r}\]
with
- \(E\) the bail free energy between the charges and
- \(q_1\) and \(q_2\) are the charges of the interacting ions and
- \(r\) is the altitude separating them.
This approach leads to the correct prediction that large cations of low charge, such as \(Thou^+\) and \(Na^+\), should class few coordination compounds. For transition metal cations that contain varying numbers of d electrons in orbitals that are Not spherically symmetric, notwithstanding, the situation is quite different. The shapes and occupations of these d-orbitals then become important in building an accurate description of the bond energy and properties of the transition element compound.
When examining a single transition metal ion, the five d-orbitals take the same energy (Figure \(\PageIndex{1}\)). When ligands approach the metal ion, some experience more than opposition from the d-orbital electrons than others based on the geometric construction of the molecule. Since ligands approach from dissimilar directions, not all d-orbitals interact directly. These interactions, nevertheless, create a splitting due to the electrostatic environment.
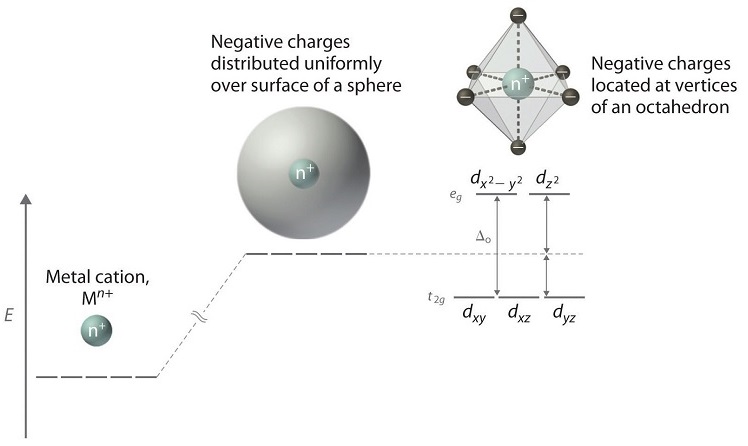
For example, consider a molecule with octahedral geometry. Ligands approach the metal ion forth the \(x\), \(y\), and \(z\) axes. Therefore, the electrons in the \(d_{z^2}\) and \(d_{10^two-y^2}\) orbitals (which prevarication along these axes) feel greater repulsion. It requires more energy to have an electron in these orbitals than it would to put an electron in ane of the other orbitals. This causes a splitting in the free energy levels of the d-orbitals. This is known as crystal field splitting. For octahedral complexes, crystal field splitting is denoted by \(\Delta_o\) (or \(\Delta_{oct}\)). The energies of the \(d_{z^2}\) and \(d_{x^two-y^2}\) orbitals increase due to greater interactions with the ligands. The \(d_{xy}\), \(d_{xz}\), and \(d_{yz}\) orbitals decrease with respect to this normal energy level and get more stable.
Electrons in Orbitals
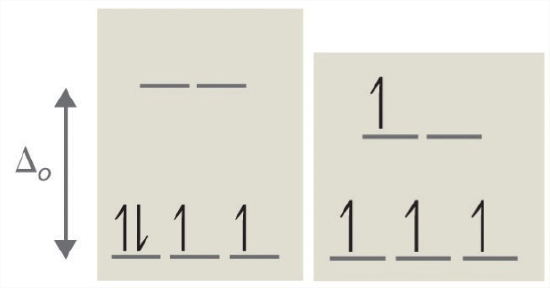
According to the Aufbau principle, electrons are filled from lower to higher energy orbitals (Figure \(\PageIndex{i}\)). For the octahedral case higher up, this corresponds to the dxy, dxz, and dyz orbitals. Following Hund'southward rule, electrons are filled in club to have the highest number of unpaired electrons. For example, if ane had a dthree complex, there would be iii unpaired electrons. If one were to add an electron, yet, it has the ability to fill up a higher free energy orbital ( d z² or d x²-y² ) or pair with an electron residing in the d xy , d xz , or d yz orbitals . This pairing of the electrons requires energy (spin pairing energy). If the pairing energy is less than the crystal field splitting energy, ∆₀, then the next electron volition go into the d xy , d xz , or d yz orbitals due to stability. This situation allows for the least amount of unpaired electrons, and is known as depression spin. If the pairing energy is greater than ∆₀, then the next electron will become into the d z ² or d x ²- y ² orbitals as an unpaired electron. This state of affairs allows for the most number of unpaired electrons, and is known as high spin. Ligands that cause a transition metal to accept a small crystal field splitting, which leads to high spin, are called weak-field ligands. Ligands that produce a large crystal field splitting, which leads to low spin, are called strong field ligands.
As mentioned in a higher place, CFT is based primarily on symmetry of ligands effectually a central metal/ion and how this anisotropic (properties depending on direction) ligand field affects the metal's atomic orbitals; the energies of which may increase, subtract or non be affected at all. One time the ligands' electrons interact with the electrons of the d-orbitals, the electrostatic interactions crusade the free energy levels of the d-orbital to fluctuate depending on the orientation and the nature of the ligands. For case, the oxidation state and the strength of the ligands decide splitting; the college the oxidation state or the stronger the ligand, the larger the splitting. Ligands are classified equally strong or weak based on the spectrochemical serial:
I- < Br- < Cl- < SCN- < F- < OH- < oxtwo-< ONO- < HiiO < SCN - < EDTAfour - < NH3 < en < NO2 - < CN-
Notation that SCN- and NO2- ligands are represented twice in the above spectrochemical series since there are two unlike Lewis base sites (e.g., gratis electron pairs to share) on each ligand (e.chiliad., for the SCN- ligand, the electron pair on the sulfur or the nitrogen tin grade the coordinate covalent bond to a metallic). The specific atom that binds in such ligands is underlined.
In addition to octahedral complexes, two common geometries observed are that of tetrahedral and square planar. These complexes differ from the octahedral complexes in that the orbital levels are raised in energy due to the interference with electrons from ligands. For the tetrahedral circuitous, the dxy, dxz, and dyz orbitals are raised in energy while the dz², dx²-y² orbitals are lowered. For the square planar complexes, there is greatest interaction with the dx²-y² orbital and therefore it has higher energy. The next orbital with the greatest interaction is dxy, followed below past dz². The orbitals with the lowest energy are the dxz and dyz orbitals. At that place is a large free energy separation betwixt the dz² orbital and the dxz and dyz orbitals, meaning that the crystal field splitting energy is large. We discover that the square planar complexes have the greatest crystal field splitting energy compared to all the other complexes. This ways that most square planar complexes are low spin, potent field ligands.
Description of d-Orbitals
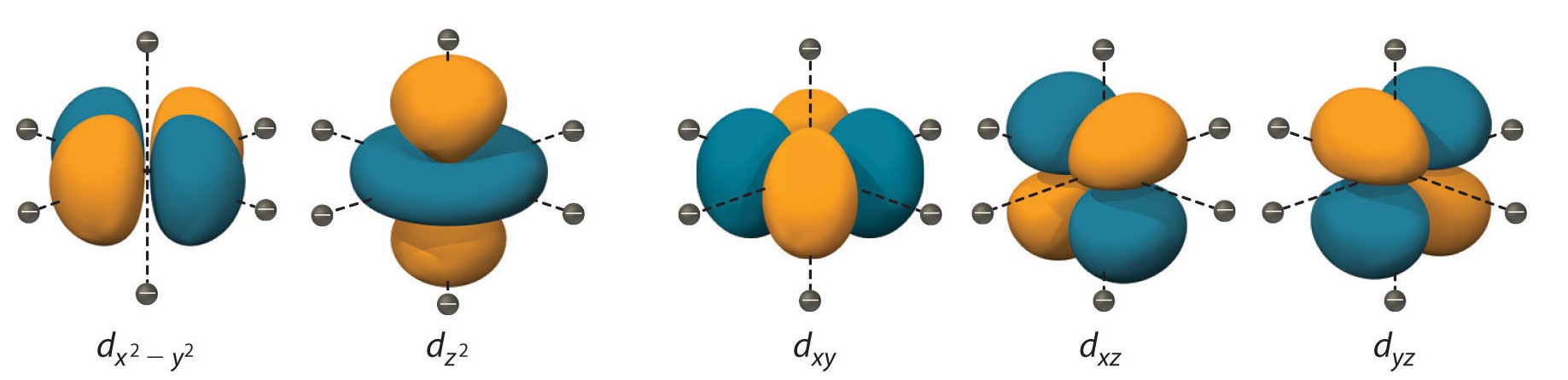
To understand CFT, one must understand the description of the lobes:
- dxy: lobes lie in-between the 10 and the y axes.
- dxz: lobes lie in-between the x and the z axes.
- dyz: lobes lie in-between the y and the z axes.
- dxtwo-ytwo : lobes lie on the ten and y axes.
- dz 2 : at that place are two lobes on the z axes and there is a donut shape ring that lies on the xy aeroplane around the other two lobes.
Octahedral Complexes
In an octahedral complex, there are six ligands attached to the central transition metal. The d-orbital splits into two dissimilar levels (Effigy \(\PageIndex{4}\)). The lesser three energy levels are named \(d_{xy}\), \(d_{xz}\), and \(d_{yz}\) (collectively referred to as \(t_{2g}\)). The two upper energy levels are named \(d_{x^²-y^²}\), and \(d_{z^²}\) (collectively referred to as \(e_g\)).
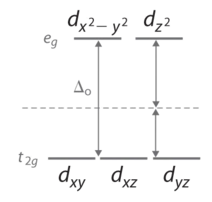
The reason they split up is because of the electrostatic interactions betwixt the electrons of the ligand and the lobes of the d-orbital. In an octahedral, the electrons are attracted to the axes. Whatever orbital that has a lobe on the axes moves to a higher energy level. This means that in an octahedral, the energy levels of \(e_g\) are higher (0.six∆o) while \(t_{2g}\) is lower (0.4∆o). The distance that the electrons have to motility from \(t_{2g}\) from \(e_g\) and it dictates the energy that the complex will absorb from white light, which will determine the color. W hether the complex is paramagnetic or diamagnetic will be determined past the spin state. If there are unpaired electrons, the complex is paramagnetic; if all electrons are paired, the complex is diamagnetic.
Tetrahedral Complexes
In a tetrahedral complex, there are four ligands fastened to the central metal. The d orbitals also split into two different energy levels. The top three consist of the \(d_{xy}\), \(d_{xz}\), and \(d_{yz}\) orbitals. The bottom 2 consist of the \(d_{x^ii-y^2}\) and \(d_{z^ii}\) orbitals. The reason for this is due to poor orbital overlap betwixt the metal and the ligand orbitals. The orbitals are directed on the axes, while the ligands are not.
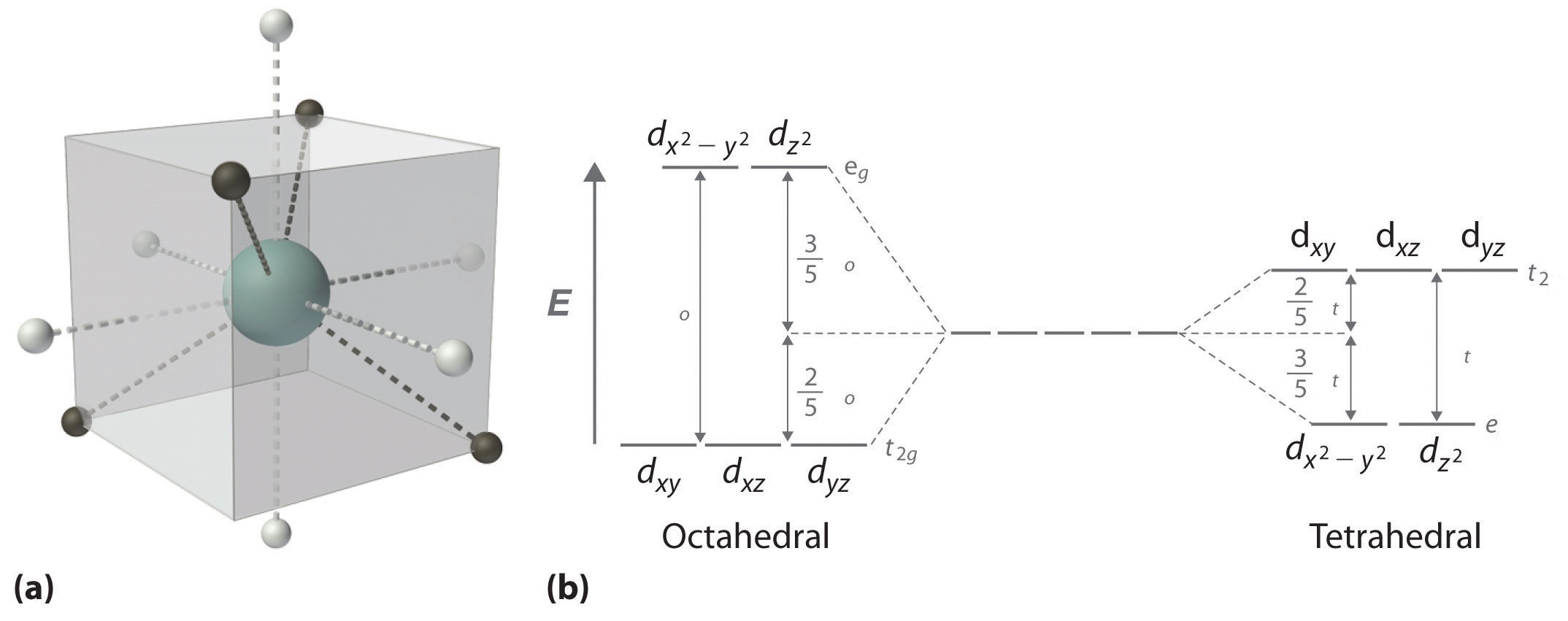
The difference in the splitting energy is tetrahedral splitting constant (\(\Delta_{t}\)), which less than (\(\Delta_{o}\)) for the same ligands:
\[\Delta_{t} = 0.44\,\Delta_o \label{1}\]
Consequentially, \(\Delta_{t}\) is typically smaller than the spin pairing energy, so tetrahedral complexes are usually high spin.
Square Planar Complexes
In a square planar, at that place are iv ligands as well. However, the departure is that the electrons of the ligands are just attracted to the \(xy\) plane. Any orbital in the xy plane has a higher energy level (Figure \(\PageIndex{6}\)). There are four different energy levels for the square planar (from the highest energy level to the lowest energy level): dten2 -y2 , dxy, dztwo , and both dxz and dyz.
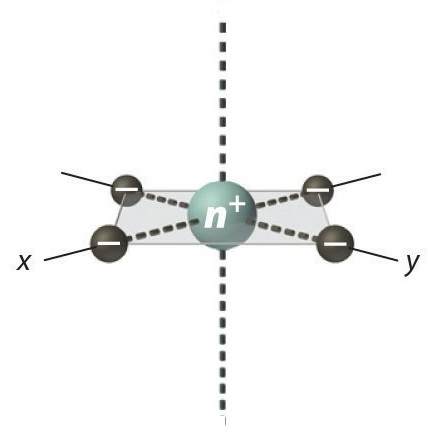
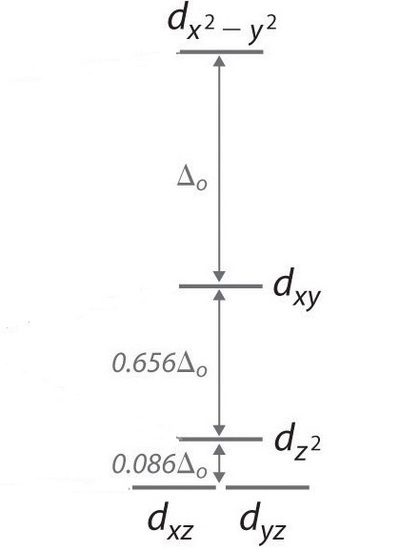
The splitting energy (from highest orbital to lowest orbital) is \(\Delta_{sp}\) and tends to exist larger then \(\Delta_{o}\)
\[\Delta_{sp} = 1.74\,\Delta_o \label{2}\]
Moreover, \(\Delta_{sp}\) is also larger than the pairing energy, and so the square planar complexes are usually low spin complexes.
Example \(\PageIndex{1}\)
For the complex ion [Fe(Cl)six]3- determine the number of d electrons for Iron, sketch the d-orbital free energy levels and the distribution of d electrons among them, list the number of lonely electrons, and characterization whether the complex is paramagnetic or diamagnetic.
Solution
- Step 1: Determine the oxidation country of Fe. Here it is Fe3 +. Based on its electron configuration, Feiii+ has v d-electrons.
- Pace 2: Determine the geometry of the ion. Here information technology is an octahedral which means the energy splitting should look similar:

- Step 3: Determine whether the ligand induces is a potent or weak field spin by looking at the spectrochemical series. Cl- is a weak field ligand (i.e., information technology induces high spin complexes). Therefore, electrons fill all orbitals before being paired.
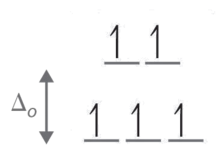
- Footstep 4: Count the number of alone electrons. Here, there are 5 electrons.
- Step five: The five unpaired electrons means this complex ion is paramagnetic (and strongly and so).
Example \(\PageIndex{2}\)
A tetrahedral complex absorbs at 545 nm. What is the respective octahedral crystal field splitting (\(\Delta_o\))? What is the color of the complex?
Solution
\[\begin{align*} \Delta_t &= \dfrac{hc}{\lambda}\\[4pt] &= \dfrac{ (6.626 \times 10^{-34} J \cdot s)(3 \times 10^viii m/s)}{545 \times 10^{-9} m} \\[4pt] &=3.65 \times 10^{-xix}\; J \end{align*}\]
However, the tetrahedral splitting (\(\Delta_t\)) is ~4/ix that of the octahedral splitting (\(\Delta_o\)).
\[\begin{align*} \Delta_t &= 0.44\Delta_o \\[4pt] \Delta_o &= \dfrac{\Delta_t}{0.44} \\[4pt] &= \dfrac{three.65 \times 10^{-19} J}{0.44} \\[4pt] &= 8.xxx \times 10^{-18}J \end{align*}\]
This is the energy needed to promote i electron in one complex. Ofttimes the crystal field splitting is given per mole, which requires this number to be multiplied by Avogadro's Number (\(6.022 \times 10^{23}\)).
This complex appears crimson, since it absorbs in the complementary green color (determined via the color wheel).
Video:
Problems
For each of the following, sketch the d-orbital free energy levels and the distribution of d electrons amid them, state the geometry, list the number of d-electrons, list the number of lone electrons, and label whether they are paramagnetic or dimagnetic:
- [Ti(H2O)6]2+
- [NiCl4]2-
- [CoF6]3- (too land whether this is low or high spin)
- [Co(NHiii)six]three+ (also state whether this is depression or high spin)
- Truthful or False: Square Planer circuitous compounds are usually depression spin.
Answers
ane. octahedral, two, two, paramagnetic
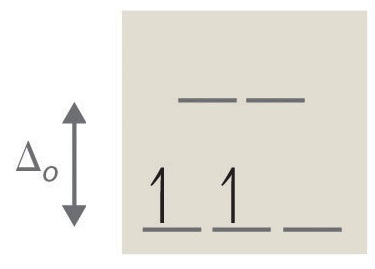
2. tetrahedral, 8, two, paramagnetic (run into Octahedral vs. Tetrahedral Geometries)
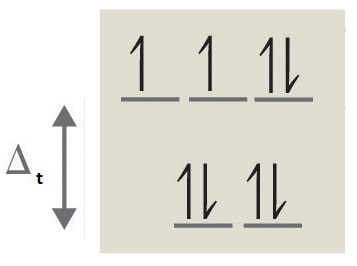
iii. octahedral, 6, 4, paramagnetic, high spin
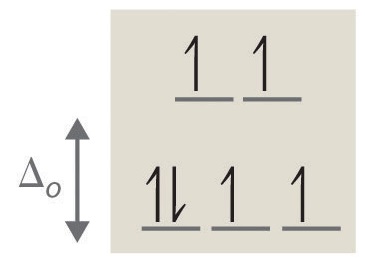
4. octahedral, 6, 0, diamagnetic, low spin
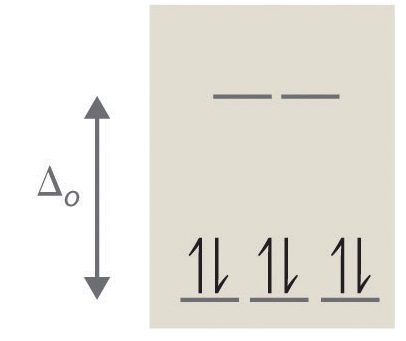
5. True
Contributors and Attributions
- Asadullah Awan (UCD), Hong Truong (UCD)
-
Prof. Robert J. Lancashire (The Department of Chemical science, Academy of the Due west Indies)
Source: https://chem.libretexts.org/Bookshelves/Inorganic_Chemistry/Supplemental_Modules_and_Websites_(Inorganic_Chemistry)/Crystal_Field_Theory/Crystal_Field_Theory
Posted by: kimblenovence.blogspot.com


0 Response to "How To Draw Crystal Field Splitting Diagram"
Post a Comment